What is pH METER OR Analyzers and explanation
In which industry we are using pH meter and how much useful for those industries. pH meter are essential and important for many industries like petrochemical plants, food processing companies, power plants, sewage treatment plants and pharmaceutical productions.
Now What is PH?
Simple answer is, in aqueous solution, the amount of acidity and basicity or alkalinity. And in technical terms pH is negative algorithm of hydrogen ion+ concentration. How these hydrogen ions produce in solution, when some acids are added in water, it breaks water molecules may be completely or partially and give hydrogen ions in solution. The mathematical terms of pH is
pH=-log[H+]
This pH is very important of human living but to understand concept of pH you must have idea of moralities. Let us take example of how much concentration should be. Concentration of Hydrogen Ions in water 1*10-7 mole per liter. So if we take log of this value it becomes -7 which pH value.
pH value 7 it is not acidic nor basic or alkaline.
pH meter voltage:
Okay now go to the voltage how much voltage accordingly PH values.
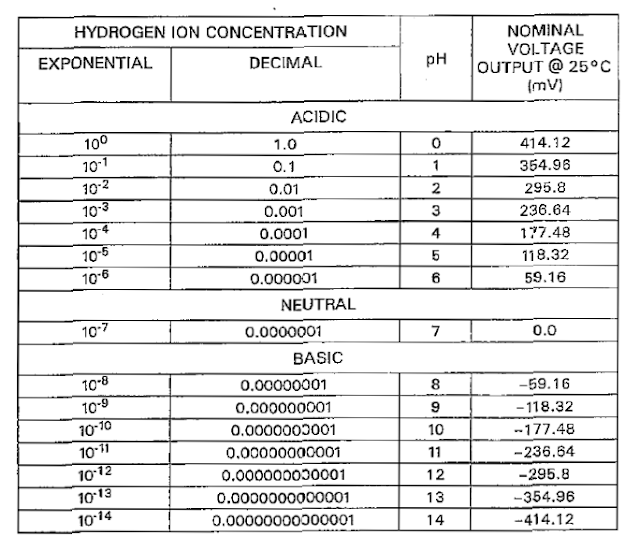
Nominal voltage output @ 250c of PH 7.0 is 0 mvolts.
Nominal voltage output @ 250c of PH 4.0 is 177.48 mvolts.
Nominal voltage output @ 250c of PH 10 is -177.48 mvolts.
How PH Meter use in process instrumentation?
In instrumentation we basically use one pH meter and two electrodes. One electrode is our reference and second one for pH measuring.
What is electrode?
Electrode made up of thin fragile glass which is used for sensing the hydrogen ions at its tip.
How electrode produce constant voltage?
When potential difference of inner glass bulb and outer glass tips occurs due to concentration of hydrogen ions on its tips, potential difference across electrode develops.
pH meter compares both signal which is received from reference electrode and measuring electrode and amplify the signal and changes to pH value. If the voltage is 0 m volts this means pH value will be 7. For each unit measurement of pH value the difference in m volts of two electrodes will be 59.16 m volts. With passage of time each pH meter need calibration because no electrode is perfect for long time.
What thing can affect the pH meter?
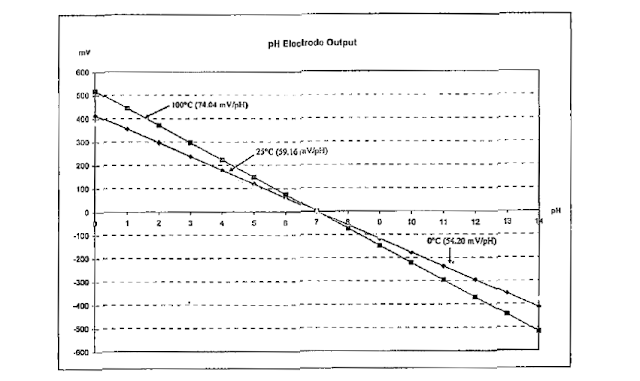
That’s the temperature it is very important for measuring pH. In pH meter two type of temperature compensation available. In advanced and new technology based pH meter has ATC Automatic temperature compensation option in meter which does automatic temperature compensation. If ATC is not available then we can adjust wit thermometer, temperature dial, some type of temperature on pH meter.
Temperature chart shows the deviation of electrode at 00c and at 250c. please see the difference.
Protection of pH Electrode
How to PH electrode we can protect if we are not using in field.
Make sure our electrode not kept in deionized water for long time. Must be protect and store in special bottles.
Most important point is store in buffer solution having 4.0 PH or 3.8 MKCl solution.
If you will not take above precaution, may be it will dry out and breaks after some time.
x
Nice blog…
Thanks for Sharing…
Humidity Meter, Countroller, pH Meter, Data Logger