Following topics to be discussed here.
- SCOPE
- REFERENCES
- GENERAL
- SAFETY
- APPLICATIONS
- JOINT DESIGN
- JOINT PREPARATION
- SOLDERS
- FLUXES
- SOLDERING METHODS AND TECHNIQUES
- CLEANUP
- INSPECTIONS
FIGURE
1 – Use of Tube Plug
TABLE
I – Solders
- IA. Lead Free Solders
- IB. Tin – Lead Solders
- IC. Tin – Silver Solders
- ID. Tin – Zinc Solders
- IE. Lead – Silver Solders
- IF. Zinc – Aluminum Solders
- IG. Tin – Antimony Solders
II – Solder – Flux Requirements
Soft Soldering Procedure
1. Scope
This Article is about solders and fluxes melting below 427 C (800 F), joint design, application temperature, testing, and soldering equipment.
2. References
Reference is made in this standard to the following documents.
Occupational Safety and Health Administration (OSHA) of U.S.A.
1910 Code of Federal Regulation Part 1910
1926 Code of Federal Regulation Part 1926
3. General
3.1 Soldering is the joining of two metal surfaces using a nonferrous filler metal (solder) that melts at a temperature below that of the metals to be joined. The melting temperature of soft solders is, by definition, below 427 C (800 F). Capillary (wick) action distributes the solder across the joint surfaces and occurs only when the surface has been cleaned and fluxed properly and has reached the correct temperature. The strength of the joint is a function of the joint clearance, bond between solder and joined metals, and the soldered area.
3.2 Solder highly stressed materials cautiously. Cracking due to liquid metal embrittlement may occur.
3.3 See SES W03-F01 for general practices for brazing and SES W03-F02 for selection criteria for braze welding methods.
4. Safety
4.1 Safe practices prescribed in SES W02-F01 shall be followed on plant sites.
4.2 Refer to OSHA 1926.102, 1926.103, 1910.133, and 1910.134 for proper protective equipment.
5. Applications
Soldering is used in pipe and tubing joints, electrical wiring connections, fill depressions, seal sheet-metal seams, and for weight to balance small equipment.
6. Joint Design
6.1 Joint strength varies widely depending upon the solder selected.
6.2 For maximum strength, a clearance of 0.075 mm (0.0031 in) between mating parts is recommended. This provides clearance for capillary action necessary to pull the soft solder into the joint and to keep oxides from reforming.
6.3 Lap and butt are two basic joint types. The lap joint design is recommended. Butt joints shall be used only where space and weight are critical.
7. Joint Preparation
7.1 Wetting / Flowing. The joint surfaces shall be mechanically and chemically protected with a flux during soldering.
7.2 Mechanical Cleaning. The following methods can be used for removing foreign material:
a. Sanding or grinding
b. Hand filing or sanding
c. Wirebrushing or scraping d. Shotblasting
7.3 Chemical Cleaning. Mechanically cleaned metal surfaces exposed to air react and form oxide films. Aluminum, magnesium, and stainless steel form refractory oxide films. Copper and silver form easily dissolved oxides. Refractory oxides require use of a corrosive flux. Softer oxides require only a less corrosive material.
8. Solders
8.1 Except for potable (drinking) water systems, tin-lead solder (50/50) can be used for the majority of applications.
8.2 Use the lead-free solders listed in Table IA for potable water systems. For selection of the appropriate solder, see Tables I and II. The Pasty Range is the difference between the maximum temperature the alloy is solid and the minimum temperature it is liquid.
9. Fluxes
9.1 After mechanically cleaning the metal, fluxes are used to prevent oxides from reforming thus enhancing flow during soldering. Fluxes can be a liquid or paste, corrosive or noncorrosive. Use the mildest flux that will adequately remove and exclude oxides from the joint being soldered. See Table II.
9.2 The disadvantage of corrosive (inorganic and organic) fluxes is that they produce a residue. If not removed or neutralized, they remain active after the joint is soldered, thus corroding the adjacent areas. Chloride containing fluxes may cause stress corrosion cracking when used on stainless steel material. Phosphoric acid type fluxes can avoid the stress corrosion problem. Their use is limited to below 260 C (550 F). For reasons of the residue problem, corrosive fluxes shall not be used on delicate electronic work and instruments.
9.3 There are three basic types of noncorrosive fluxes: nonactive rosin, mildly activated rosin, and activated rosin. Rosin fluxes shall not be used with flame heating as they will decompose without cleaning effectively.
10. Soldering Methods and Techniques
10.1 After the surfaces to be joined are properly cleaned, apply sufficient flux to completely cover the joint surface(s). The joint is now ready for the application of heat either by soldering irons (copper bit or quick-heating gun type) or by flame.
10.2 Soldering irons consist of a copper alloy bit that is electrically or flame-heated. The bit conducts heat readily and is easily ‘tinned’ (coated with solder). The temperature of the iron shall be maintained between 371-399 C (700-750 F) for efficient soldering. Temperatures in excess of 399 C (750 F) will cause the bit to scale introducing tinning problems (tin will burn out of tin-lead solder resulting in poor solder adhesion) and cause rosin fluxes to carbonize.
10.2.1 Apply the soldering iron to the metal surface(s) to obtain maximum contact surface of the tip. When the melting temperature of the solder is reached, the solder is placed on the joint surface. The melted solder will flow onto or between the joint surfaces. Melting of flux-core solder on the tip of the soldering iron is not recommended as the action of the flux is destroyed.
10.2.2 To maintain maximum soldering efficiency, the tip of the soldering iron shall be kept clean of scale and well coated with solder. Periodically wipe the tip with steel wool or rub on a cake of salammoniac. A damp sponge can also be used to clean soldering iron tips.
10.3 Soldering guns heat up due to large current passing through a copper loop which is connected to the secondary of a step-down transformer. A copper bit is fastened to the tip of the copper loop. These quick heating solder guns are suitable for light intermittent work.
10.4 Flame heating is used extensively in soldering copper pipe and tubing joints. As the solder starts to flow, the flame is removed and the solder wire is fed into the joint. The length of solder wire consumed will be approximately equal to the diameter of the joints. The connection is completed by wiping off excess solder with a clean cloth.
10.4.1 The presence of moisture results in an incomplete solder joint. Special tools are available to control liquid flow.
10.4.1.1 Piping or tubing joint(s) shall be isolated from the system and allowed to drain. One simple means of preventing residual water from draining into the joint area is to place a plug of bread upstream of the joint and proceed with the soldering. After the solder joint is complete and cooled, the water supply is turned on. The bread will soften and flow out. If the introduction of a foreign material will cause problems to the system, then other means of stopping the drainage shall be used, for example, using dry ice or liquid nitrogen to freeze the line or using a ‘soldering aide’.
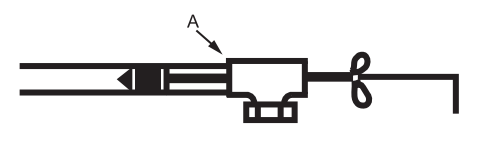
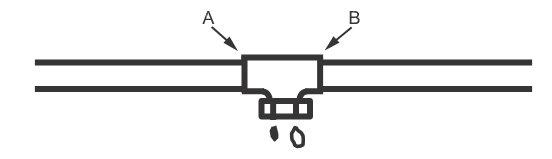
10.4.1.2 The use of tube plugs prevent intermittent drainage. Tube plugs, available in sizes from 12.7 to 76.2 mm (1/2 to 3 in), are inserted through an inverted drain tee and into the copper tubing to be joined. The tool is expanded to stop any drainage and the drain tee is soldered to the copper tubing. After this joint is made, the plug is removed and subsequent joints can be soldered downstream of the drain tee without moisture problems. See Figure 1.
11. Cleanup
11.1 The harmful effect of corrosive fluxes on the jointed material requires that they shall be removed. Corrosive fluxes absorb water and form a corrosive mixture.
11.2 Corrosive fluxes can be removed with mineral spirits or solvents.
11.3 Corrosive fluxes can be removed by cleaning the soldered joint with a damp rag. The usual practice is to do nothing on soldered plumbing.
11.4 Noncorrosive fluxes can be removed with solvents.
12. Inspections
12.1 The following is a brief description of common soldered joint conditions:
a. Good Solder Joint. This is shiny and smoot
b. Cold Solder Joint. This defect is caused by insufficient heating of the joint. Heat shall be applied to the metal, not the solder. A cold solder joint usually will appear chalky or crystalline, or may show as unbonded solder droplets.
c. Rosin Joint. This defect arises from insufficient heat (too small an iron or too short a contact time) and results in an interface of rosin between the joint surfac
d. Disturbed Solder. In this joint, one or more of the parts moved before the solder completely solidifies. Parts shall be held rigid during the soldering operation.
e. Solder Shorts. A solder short will develop if excess solder is allowed to bridge between adjoining electrical conductors. Where space limitations are tight, both the soldering iron and the solder wire diameter should be sma
f. Insufficient Solder. These joints are weak and may result in joint failur
TABLE I
Solders
Table IA. Lead Free Solders
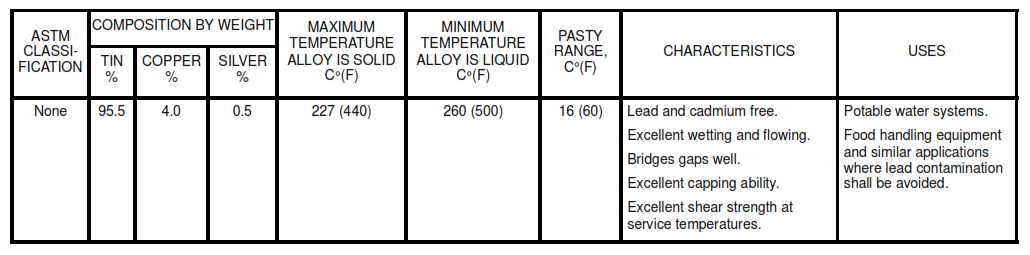
Table – IB . Tin – Lead Solders
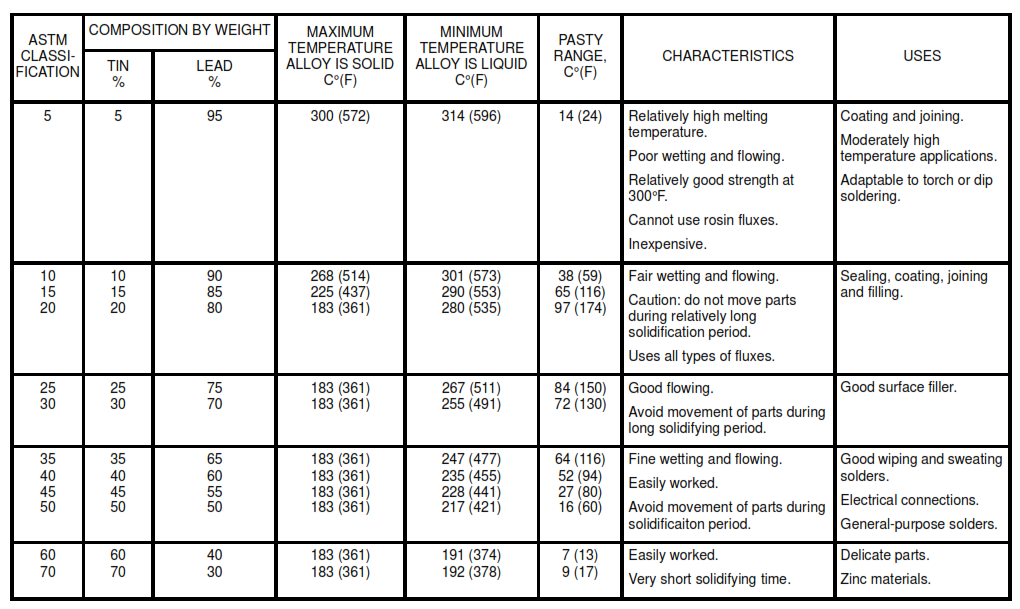
Table – IC . Tin – Silver Solders
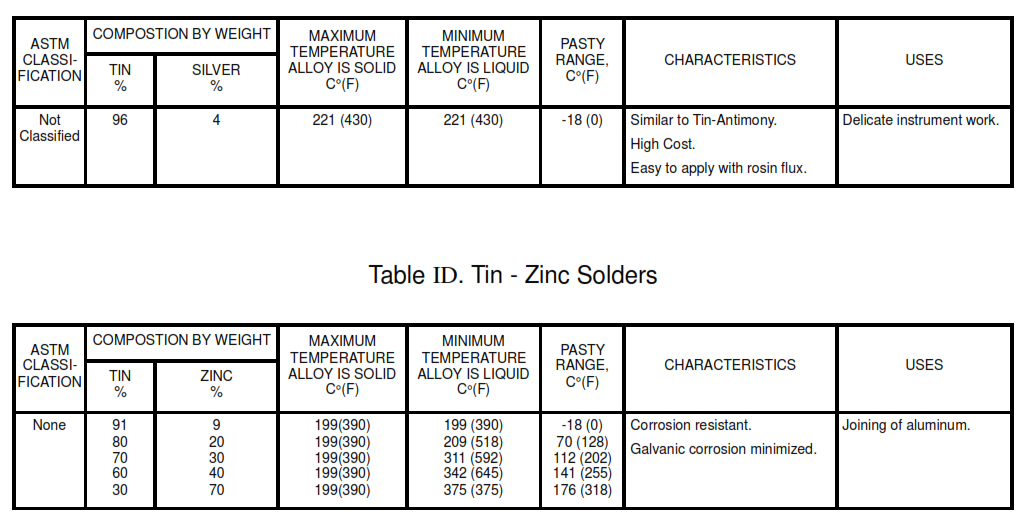
Table IE. Lead – Silver Solders
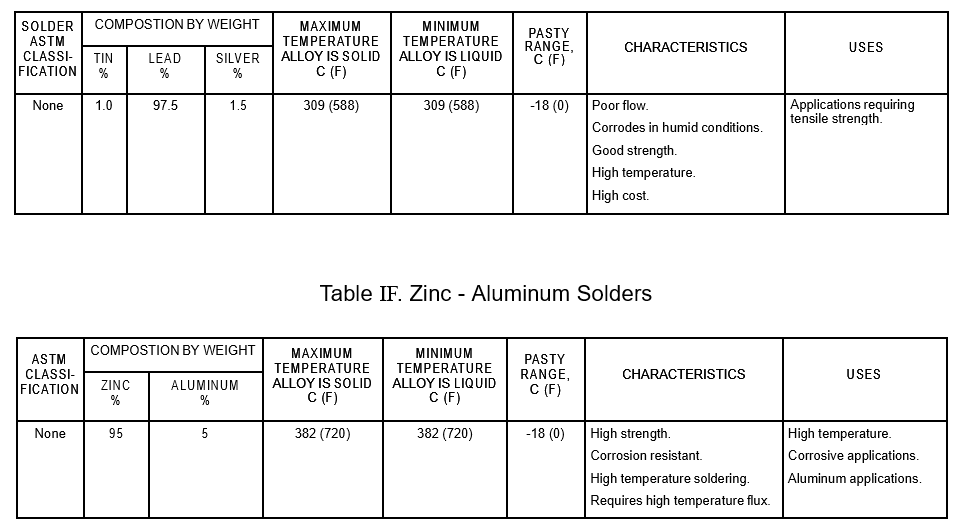
Table IG. Tin – Antimony Solders
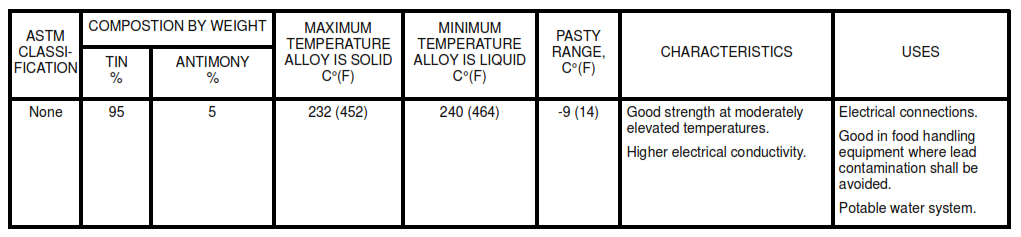
TABLE II Solder – Flux Requirements
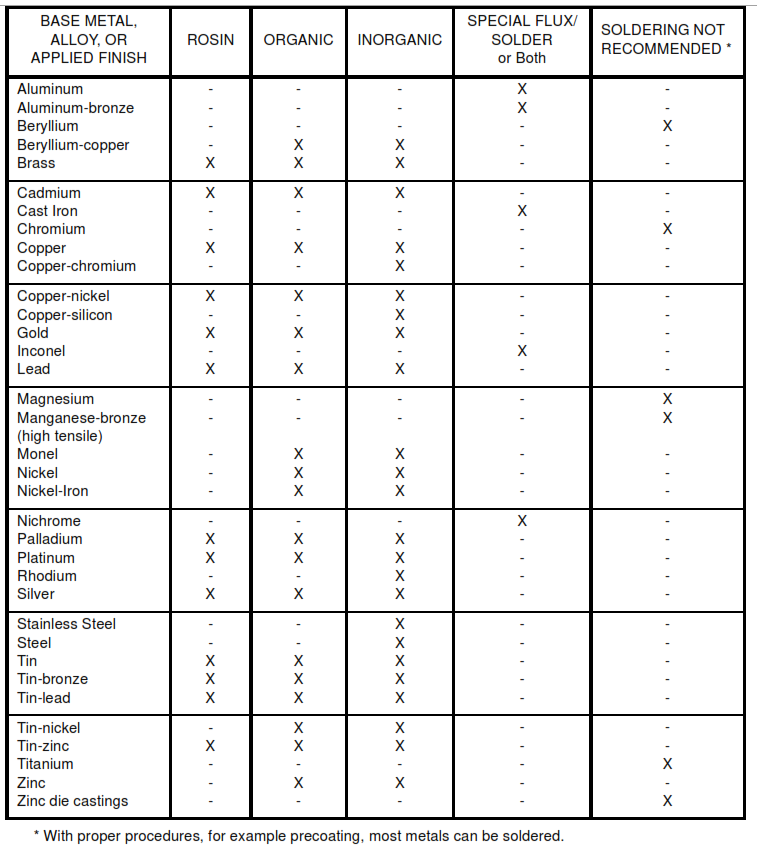
FIGURE 1 – Use of Tube Plug
Step 1. Slip an inverted clean out T on to the copper service. Insert and expand proper size plug. Solder at A. Water is held back by Soldering Aide.
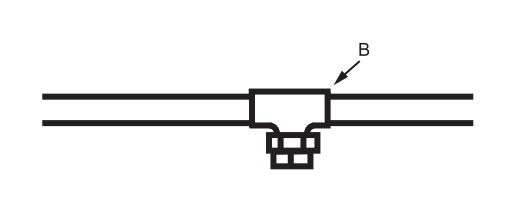
Step 2. After soldering at A remove plug and solder at B. Water will run out inverted clean out T and will not affect any solder joints beyond point B.
Step 3. After completing job, beyond point B, pipe dope the clean out plug and insert in clean out T.